|
11/19/2023 0 Comments Lattice energy of nacl 8 
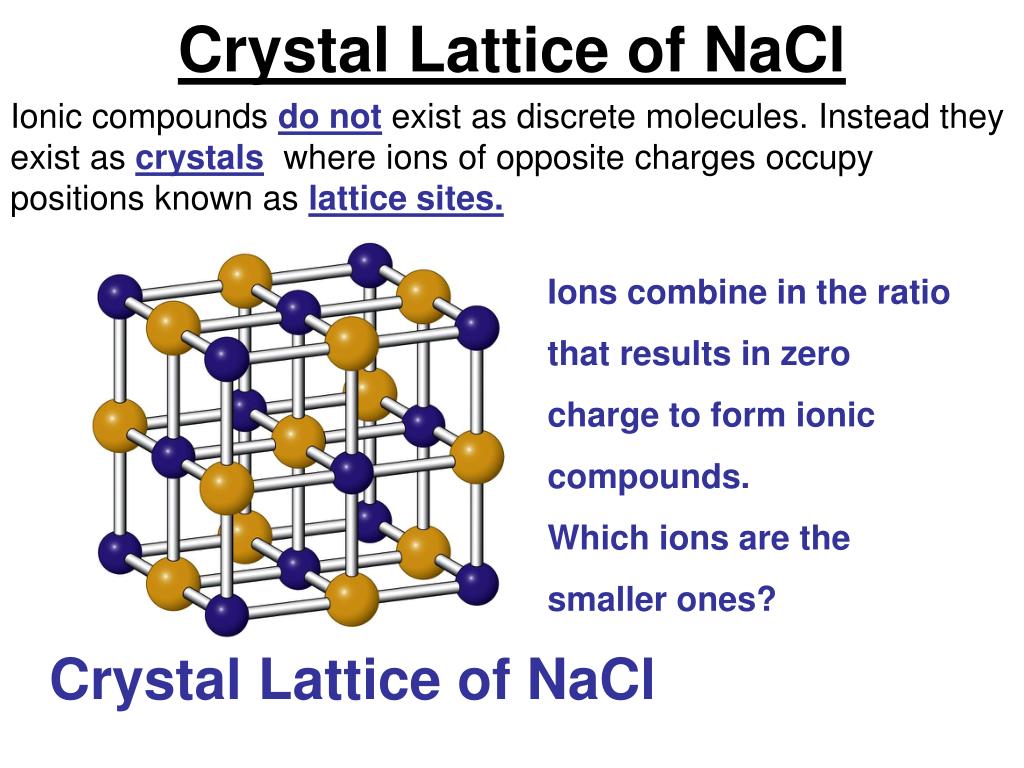
Na + (g) + Cl –(g) → NaCl (s) ΔH LE = ? Determining Lattice Energy from Hess’ Lawįrom Hess’s Law, the overall change in enthalpy of a process can be determined by breaking the process down into steps and then adding the enthalpy changes of each step. 
The gaseous sodium ion and gaseous chloride ion combine to form a solid sodium chloride molecule and release energy (exothermic) equivalent to the lattice energy (ΔH LE), which we shall determine. The last step is forming the ionic compound from its constituent gaseous ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
